[:pb]Authors: Cardoso, L. N. F.; Nogueira, T. C. M.; Wardell, J. L.; De Souza, M. V. N.; Harrison, W. T. A.
Source: Acta Crystallographica Section e, Crystallographic Communications, v. 74, p. 313-318, 2018
Publisher: Science Direct
Abstract
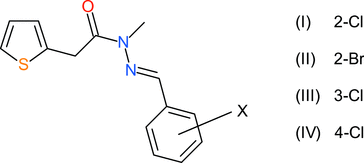
The crystal structures of three isomeric (E)-N′-(chlorophenylmethylidene)-N-methyl-2-(thiophen-2-yl)acetohydrazides (C14H13ClN2OS) are described, with the Cl atom in ortho (I), meta (III) and para (IV) positions in the benzene ring. The ortho-bromo derivative (II) (C14H13BrN2OS), which is isostructural with its chloro congener (I), is also reported. Molecules (I)–(III) have similar conformations, which approximate to L-shapes, as indicated by their N—C—C—Ct (t = thiophene) torsion angles of −90.1 (3), −91.44 (18) and −90.7 (9)°, respectively. The conformation of (IV) is different, with an equivalent torsion angle of −170.75 (11)° corresponding to a more extended shape for the molecule. The thiophene ring in each structure features `flip’ rotational disorder. The packing for (I) and (II) features inversion dimers, linked by pairs of C—H O interactions, which generate R22(14) loops. In the crystal of (III), [010] C(8) chains arise, with adjacent molecules linked by pairs of C—H
O interactions, which generate R22(14) loops. In the crystal of (III), [010] C(8) chains arise, with adjacent molecules linked by pairs of C—H O hydrogen bonds. The packing for (IV) features unusually short C—H
O hydrogen bonds. The packing for (IV) features unusually short C—H O interactions arising from an H atom attached to the benzene ring (H
O interactions arising from an H atom attached to the benzene ring (H O = 2.18 Å), which lead to C(9) [301] chains. Hirshfeld fingerprint percentage contact contributions are similar for the four title compounds.
O = 2.18 Å), which lead to C(9) [301] chains. Hirshfeld fingerprint percentage contact contributions are similar for the four title compounds.
Keywords: crystal structure,carbohydrazide,methylation,weak hydrogen bonds
Document Type: Research Article
DOI: 10.1107/S2056989018001251
Publication date: Março de 2018[:]