A substitution reaction known for destroying stereochemistry has been converted into a chirality-creating reaction to tackle one of the toughest organic chemistry challenges: making quaternary carbon centres.
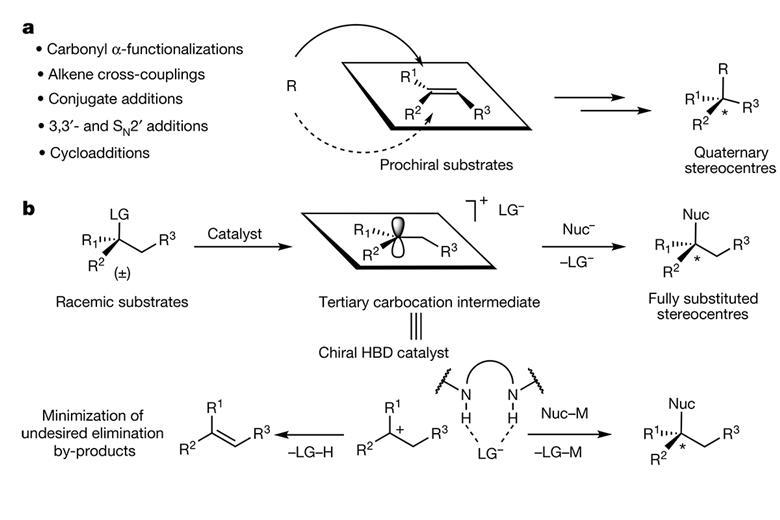
There is currently no perfect way to make quaternary stereocentres. Most methods rely on using a pro-chiral substrate – a compound like a tri-substituted alkene with well-defined stereochemistry. But making these pro-chiral substrates is a challenge in itself. Eric Jacobsen from Harvard University, US, came up with an unusual idea: he wanted to control stereochemistry during the SN1 reaction, a nucleophilic substitution every student knows from their introductory organic chemistry course. It creates a flat carbocation intermediate that Jacobsen thought could act as the pro-chiral substrates. However, the SN1 reaction is also known for destroying stereochemistry. Since a nucleophile can approach the carbocation from either side, traditional SN1 reactions produce racemates, even if the starting material is a single enantiomer. ‘I would talk about the SN1 mechanism to my students and I’d say “Wouldn’t it be interesting if you could actually control the delivery of the nucleophile back into this carbocation intermediate?”’ Jacobsen recalls. ‘Then, instead of losing the stereochemistry, you could create it.’ Jacobsen and his team have now designed a catalytic SN1 reaction that converts a racemic mixture into a chiral quaternary centre. Starting off with a racemic mixture of propargyl acetate, the chiral catalyst removes the acetoxy group and exchanges it for an allyl group, creating a single enantiomer.